Natural killer (NK) cells are central to innate immune defense and have two important functions in the body. Firstly to kill virus-infected and tumor cells and secondly to interact with other cells as part of the immune response process. NK cells effectively recognize virus-infected cells and tumor cells, making NK cells an important part of the body’s protection against cancer.
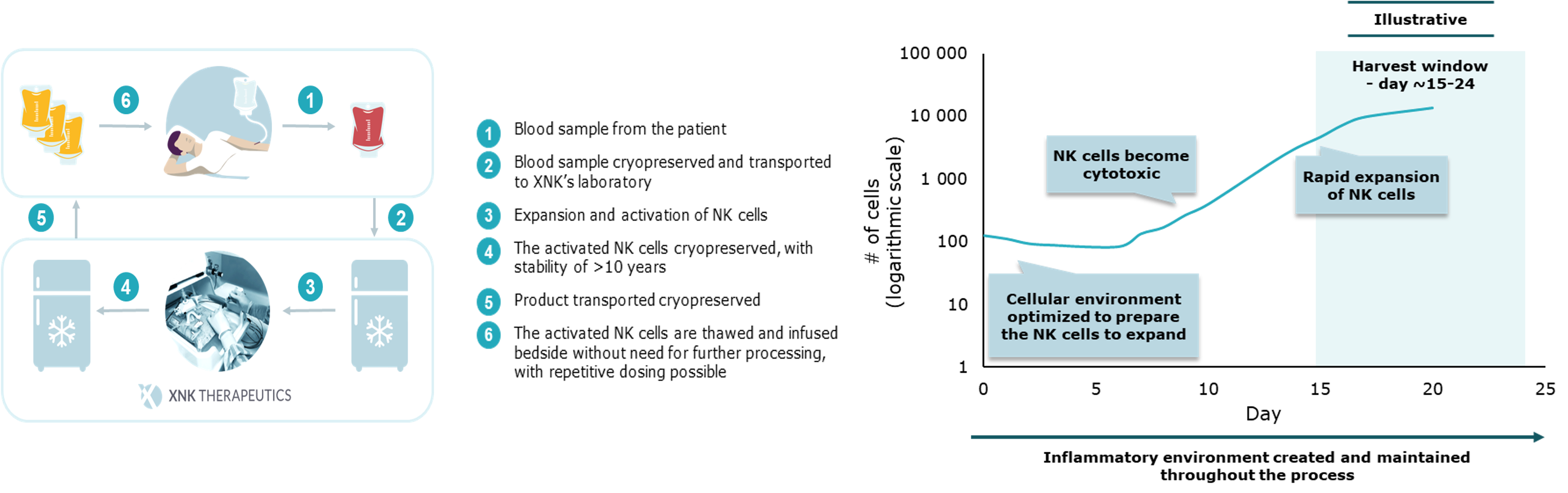
XNK has developed its own proprietary technology platform and the autologous NK cell therapy drug candidate evencaleucel, to target cancer and especially in situations where autologous therapies have clear benefits over allogeneic cell products. The platform enables selective expansion and activation of NK cells from the peripheral blood of cancer patients to produce NK cells with increased tumor killing capacity.
The expansion and activation of the patient’s NK cells take less than three weeks and the product is delivered to the clinic on demand, where it is thawed and infused to the patient without any further processing. The system is feeder cell free and both starting material and final product can be frozen, resulting in a robust and cost-efficient process. The patented technology and investigational drug product evencaleucel have been tested in a successful first-in-human phase I clinical trial and phase II studies are ongoing.
Autologous vs. Allogeneic NK cells
Autologous cell therapies are truly personalized therapies where the patient’s own blood provides the starting material from which a unique and individualized product is produced. An autologous product is therefore not an ‘off-the-shelf’ or ‘one-size-fits-all’ product and robust and cost-efficient manufacturing processes and logistics are essential.
One advantage of autologous NK cell therapies over ‘off-the-shelf’ allogeneic NK cells therapies is that they can be infused to patients without the need for lymphodepletion (immunosuppressive treatment). Moreover, repeat dosing would not be limited with autologous cells and administration of autologous NK cells to date has demonstrated a good safety profile. Furthermore, the autologous NK cell approach could be readily applied in settings of e.g. minimal residual disease (MRD) conditions or as maintenance therapy where immunosuppressive treatment could not easily be justified. The truly individualized autologous NK cell approach should allow for an important extension of the therapeutic applications of NK cell therapy beyond what is expected to be feasible in allogeneic settings. Autologous NK cells can be expected to have application in the treatment of hematological malignancies as well as solid tumors.
Please see our position paper Characteristics & Positioning of Autologous NK Cell Therapies in Relation to Allogeneic Ditto
Broad Utility
XNK has identified several potential new oncology indications, including both hematological malignancies and solid tumors, where our product has the opportunity to provide new and much needed effective treatment, either as monotherapy or in combination with antibodies. XNK Therapeutics will continue evaluating several new potential cancer indications in preclinical studies, moving them towards the clinic, whilst concurrently conducting and planning future clinical studies in multiple myeloma (see Pipeline).